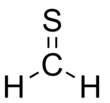
Thioformaldehyde
This compound is not observed in the condensed state (solid or liquid) because it oligomerizes to 1,3,5-trithiane, which is a stable colorless compound with the same empirical formula.
Despite the instability of these concentrated forms, thioformaldehyde as a dilute gas has been extensively studied.
For these purposes, it is produced by thermal decomposition of dimethyl disulfide.
[1] The molecule has been observed in the interstellar medium[2] and has attracted much attention for its fundamental nature.
[3] The tendency of thioformaldehyde to form chains and rings is a manifestation of the double bond rule.