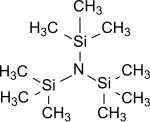
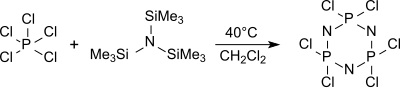Tris(trimethylsilyl)amine
[2][3][4] Early attempts to prepare tris(trimethylsilyl)amine from ammonia and trimethylchlorosilane (TMS-Cl) were unsuccessful even at temperatures of 500 °C and in the presence of the base pyridine.
[5][6] The reaction of ammonia and trimethylchlorosilane stops at the stage of the doubly silylated product bis(trimethylsilyl)amine (usually referred to as hexamethyldisilazane, HMDS).
[7] From antimony trichloride and tris(trimethylsilyl)amine, a nitridoantimone cubane-type cluster can be formed almost quantitatively at –60 °C.
[16] The cyclic trimer (NPCl2)3 hexachlorocyclotriphosphane is predominantly formed from tris(trimethylsilyl)amine and phosphorus pentachloride in boiling dichloromethane (about 40 °C) among other oligomers which gives upon heating over 250 °C high molecular weight, little defined polydichlorophosphazenes.
In chemical nitrogen fixation (i.e., the transformation of atmospheric nitrogen under normal conditions into reactive starting materials for chemical syntheses, usually also ammonia), tris(trimethylsilyl)amine plays an important role in the so-called reductive silylation, since it is hydrolyzed with water to ammonia.