Eicosanoid
Eicosanoids function in diverse physiological systems and pathological processes such as: mounting or inhibiting inflammation, allergy, fever and other immune responses; regulating the abortion of pregnancy and normal childbirth; contributing to the perception of pain; regulating cell growth; controlling blood pressure; and modulating the regional flow of blood to tissues.
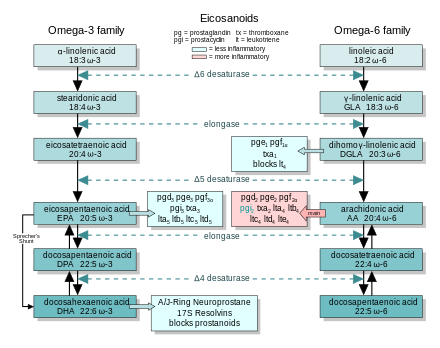
[1][2] There are multiple subfamilies of eicosanoids, including most prominently the prostaglandins, thromboxanes, leukotrienes, lipoxins, resolvins, and eoxins.
"Eicosanoid" (from Greek eicosa- 'twenty') is the collective term[8] for straight-chain PUFAs (polyunsaturated fatty acids) of 20 carbon units in length that have been metabolized or otherwise converted to oxygen-containing products.
Since eicosanoid-forming enzymes commonly make S isomer products either with marked preference or essentially exclusively, the use of S/R designations has often been dropped (e.g. 5S-HETE is 5-HETE).
Nonetheless, certain eicosanoid-forming pathways do form R isomers and their S versus R isomeric products can exhibit dramatically different biological activities.
Eicosanoid biosynthesis begins when a cell is activated by mechanical trauma, ischemia, other physical perturbations, attack by pathogens, or stimuli made by nearby cells, tissues, or pathogens such as chemotactic factors, cytokines, growth factors, and even certain eicosanoids.
The activated cells then mobilize enzymes, termed phospholipases A2 (PLA2), capable of releasing ω−6 and ω−3 fatty acids from membrane storage.
There are several classes of PLA2 with type IV cytosolic PLA2 (cPLA2) appearing to be responsible for releasing the fatty acids under many conditions of cell activation.
The enzymes that are biosynthetic for eicosanoids (e.g., glutathione-S-transferases, epoxide hydrolases, and carrier proteins) belong to families whose functions are involved largely with cellular detoxification.
[36][37] These pathways have been shown or in some cases presumed to metabolize eicosapentaenoic acid to eicosanoid analogs of the sited products that have three rather than two double bonds and therefore contain the number 3 in place of 2 attached to their names (e.g. PGE3 instead of PGE2).
Either of the latter two enzymes act to attach the sulfur of cysteine's thio- (i.e. SH) group in the tripeptide glutamate-cysteine-glycine to carbon 6 of LTA4 thereby forming LTC4.
After release from its parent cell, the glutamate and glycine residues of LTC4 are removed step-wise by gamma-glutamyltransferase and a dipeptidase to form sequentially LTD4 and LTE4.
[41][42] The decision to form LTB4 versus LTC4 depends on the relative content of LTA4 hydrolase versus LTC4 synthase (or glutathione S-transferase in cells; eosinophils, mast cells, and alveolar macrophages possess relatively high levels of LTC4 synthase and accordingly form LTC4 rather than or to a far greater extent than LTB4.
[54] The following table lists a sampling of the major eicosanoids that possess clinically relevant biological activity, the cellular receptors (see Cell surface receptor) that they stimulate or, where noted, antagonize to attain this activity, some of the major functions which they regulate (either promote or inhibit) in humans and mouse models, and some of their relevancies to human diseases.
Prostanoids also activate the PPARγ members of the steroid/thyroid family of nuclear hormone receptors, and directly influence gene transcription.
[80] Prostanoids have numerous other relevancies to clinical medicine as evidence by their use, the use of their more stable pharmacological analogs, of the use of their receptor antagonists as indicated in the following chart.
The three cysteinyl leukotrienes, LTC4, LTD4, and LTE4, are potent bronchoconstrictors, increasers of vascular permeability in postcapillary venules, and stimulators of mucus secretion that are released from the lung tissue of asthmatic subjects exposed to specific allergens.
[81] Drugs that block their activation of the CYSLTR1 receptor viz., montelukast, zafirlukast, and pranlukast, are used clinically as maintenance treatment for allergen-induced asthma and rhinitis; nonsteroidal anti-inflammatory drug-induced asthma and rhinitis (see aspirin-exacerbated respiratory disease); exercise- and cold-air induced asthma (see Exercise-induced bronchoconstriction); and childhood sleep apnea due to adenotonsillar hypertrophy (see Acquired non-inflammatory myopathy § Diet and Trauma Induced Myopathy).
[86] LxA4, LxB4, 15-epi-LxA4, and 15-epi-LXB4, like other members of the specialized pro-resolving mediators class of eicosanoids, possess anti-inflammatory and inflammation resolving activity.
They stimulate vascular permeability in an ex vivo human vascular endothelial model system,[89] and in a small study of 32 volunteers EXC4 production by eosinophils isolated from severe and aspirin-intolerant asthmatics was greater than that from healthy volunteers and mild asthmatic patients; these findings have been suggested to indicate that the eoxins have pro-inflammatory actions and therefore potentially involved in various allergic reactions.
RvE1, 18S-RvE1, RvE2, and RvE3, like other members of the specialized pro-resolving mediators) class of eicosanoids, possess anti-inflammatory and inflammation resolving activity.
Many of the 3-series prostanoids, the hydroxyeicosapentaenoic acids, and the 5-series LT have been shown or thought to be weaker stimulators of their target cells and tissues than their arachidonic acid-derived analogs.
The epoxy eicosatrienoic acids (or EETs)—and, presumably, the epoxy eicosatetraenoic acids—have vasodilating actions on heart, kidney, and other blood vessels as well as on the kidney's reabsorption of sodium and water, and act to reduce blood pressure and ischemic and other injuries to the heart, brain, and other tissues; they may also act to reduce inflammation, promote the growth and metastasis of certain tumors, promote the growth of new blood vessels, in the central nervous system, regulate the release of neuropeptide hormones, and in the peripheral nervous system inhibit or reduce pain perception.
Low dietary intake of these less-inflammatory fatty acids, especially the ω−3s, has been linked to several inflammation-related diseases, and perhaps some mental illnesses.
[95] Of these, the action on eicosanoids is the best explored Recent data in 2024 has emerged that neuronal integrity breakdown was reduced by ω−3 treatment in APOE*E4 carriers, suggesting that this treatment may be beneficial for this specific group suggested fish oil supplements might help older adults fight Alzheimer’s disease.
[97][98] In general, the eicosanoids derived from AA promote inflammation, and those from EPA and from GLA (via DGLA) are less inflammatory, or inactive, or even anti-inflammatory and pro-resolving.
Between 1929 and 1932, George and Mildred Burr showed that restricting fat from animals' diets led to a deficiency disease, and first described the essential fatty acids.