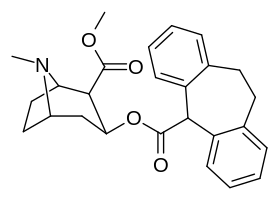
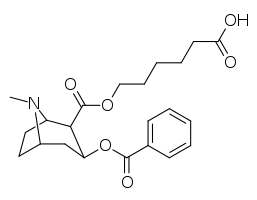
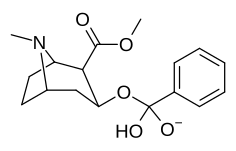
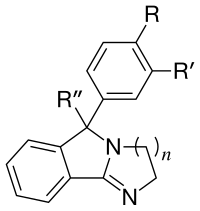
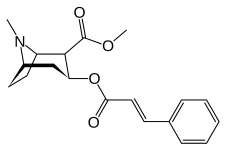
List of cocaine analogues
These include both intracellular sodium channel blocker anaesthetics and stimulant dopamine reuptake inhibitor ligands (such as certain, namely tropane-bridged-excised, piperidines).
Additionally, researchers have supported combinatorial approaches for taking the most promising analogues currently elucidated and mixing them to the end of discovering novel & efficacious compounds to optimize their utilization for differing distinct specified purposes.
[d] Due to the presence of four asymmetric carbon atoms in the 1- & 5- to 8 (N) position bond bridge that could adopt R- & S- configurations, cocaine can be considered to have as many as sixteen stereoisomers.
For example, the end product of cocaine biosynthesis contains an axial C2-carbomethoxy moiety which readily undergoes epimerization to the equatorial position via saponification.
The MAT binding pocket analogous to the lipophilic place on cocaine-like compounds, inclusive of the benzene ring, is approximate to 9 Å in length.
It is even a common case where two separate substitutions can each yield a weaker, lower affinity or even wholly non-efficacious compound respectively; but due to findings that oftentimes, when used together, such two mutually inferior changes being added in tandem to one analogue has the potential to make the resultant derivative display much greater efficacy, affinity, selectivity &/or strength than even the parent compound; which otherwise was compromised by either of those two alternations when made alone.
This finding indicates that the presence of a hydrogen bond acceptor (i.e. carbomethoxy) at the 2β position is not absolutely necessary for the creation of high affinity cocaine analogues.
In front-bridged analogs the nitrogen lone pairings rigid fixity makes it reside in an equatorial placing for the piperidine ring-part of the tropane nucleus, pointing to the two-carbon & three methylene unit bridgehead; giving the attested front-bridged cocaine analogues preference for SERT over DAT.
)[21] The difference in the length of the benzoyloxy and the phenyl linkage contrasted between cocaine and phenyltropanes makes for a shorter distance between the centroid of the aromatic benzene and the bridge nitrogen of the tropane in the latter PTs.
Despite the observation of increased stimulation, phenyltropanes lack the local anesthetic sodium channel blocking effect that the benzoyloxy imparts to cocaine.
Beside topical affect, this gives cocaine an affinity for binding to sites on the dopamine and serotonin sodium dependent transport areas that are distinct & specific to MAT in contrast to the general sodium channels; creating a separate mechanism of relational affinity to the transporters in addition to its inhibition of the reuptake for those transporters; this is unique to the local anesthetic value in cocaine & analogues with a similar substitute for the benzoyloxy that leaves the sodium channel blockage ability intact.
Rendering such compounds as different functionally in their relation to MAT contrasted to phenyltropane analogues which have the local anesthetic bridge removed.
In addition, it even has been postulated that a crucial role regarding the electron energy imparted via voltage sensitization (and thus action potential blockage with a molecule capable of intersecting its specific channel, in the case of cocaine a sodium channel, that potentially serves in re-quantifying its charge) upon a receptor binding site may attenuate the mediating influence of the inhibitory regulation that autoreceptors play by their slowing neurotransmitter release when an efflux is created through an instance of agonism by a compound; allowing said efflux to be continued without the body's attempt to maintain homeostasis enacting in as readily responsive a manner to its conformational change.
A somewhat recent occurrence among tentative modern folklore which has traversed the circling of rumors mostly confined to the likes of universities and popular culture trivia has been that cocaine is one element, or molecule increment of weight or charge etc., away from the molecular structure of sugar.
[37] Though such a statement is false as a general pretense, there is a dextrose based super-structure that has a vaguely similar overlay with cocaine which is "benzoyl-beta-D-glucoside."
uptake uptake/binding ɑInhibition at 10 μM The binding of benztropine analogues to the DAT differs significantly from that of cocaine and the phenyltropanes.
Compound 7a construed as a potentiating allosteric effect (by unveiling occluded configured serotonin uptake-area ligand-site on surface of transporter that allows for binding by exogenous ligand, when SERT is otherwise conformed in a transitional manner where a SERT ligand cannot bind, this effect with compound in question occurs) at concentrations of 10μM—30μM (wherein it acts by interconverting the conformational state of unexposed SERTs to ones exposing the SSRI binding site via a shift to the equilibrium of the MAT) while exerting an inhibitory orthosteric effect when concentrations reach >30μM and above.7a is the only known compound to allosterically modulate SERT in such a way within in vitro conditions (tianeptine has been shown to do similar, but has only shown efficacy doing so in living in vivo tissue samples).
Considering its noncompetitive inhibition of 5-HT transporters decreasing Vmax with small change in the Km for serotonin, putatively stabilizing the cytoplasm-facing conformation of SERT: in such respect it is considered to have the opposite effect profile of the anti-addiction drug ibogaine (save for the function by which its anti-addictive properties are thought to be mediated, i.e. α3β4 nicotinic channel blockage.
Similarly, such peripheral DAT considerations (when, as often is, considered conformational rather than otherwise explained as being electrostatic) may constitute the difference in affinity, through allosertic occulsion, between cyclopentyl-ruthenium phenyltropane in its difference from the tricarbonyl-chromium See: List of Mazindol analogues Mazindol is usually considered a non-habituating (in humans, and some other mammals, but is habituating for e.g. Beagles[av]) tetracyclic dopamine reuptake inhibitor (of somewhat spurious classification in the former).
[aw] In animal studies, certain of the local anesthetics have displayed residual dopamine reuptake inhibitor properties,[46] although not normally ones that are easily available.
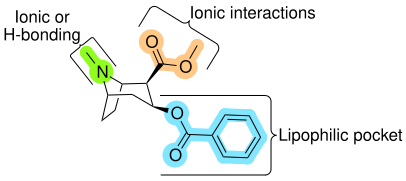
While it was originally thought that the 2β-carbomethoxy moiety interacted with the DAT through hydrogen bonding, subsequent research has indicated that electrostatic (ionic) interactions are the primary means of interactions with the DAT. [ c ]