Structural scheduling of synthetic cannabinoids
(example definition from Kentucky, which is substantially derived from the 2009 ACMD advice on synthetic cannabinoids in the UK)[11][12] This causes a substance such as MAM-2201 to be controlled as a Schedule 1 illegal drug, even though "MAM-2201" or its corresponding chemical name are not specifically listed in the statute.
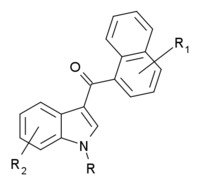
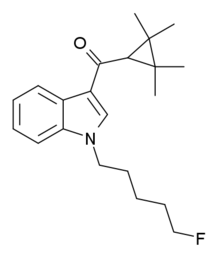
A fluoropentyl chain falls within the scope of "haloalkyl" substitutions, and so with a methyl group attached to the carbon atom at the 4-position of the naphthyl ring (i.e. "substituted in the naphthyl ring to any extent"), and a fluoropentyl group attached to the nitrogen atom ("with substitution at the nitrogen atom of the indole ring by [a]...haloalkyl...group"), this compound falls within the scope of the general definition.
On the other hand, THJ-2201 with an indazole core, falls outside this general definition, as it is a naphthoylindazole rather than a naphthoylindole.
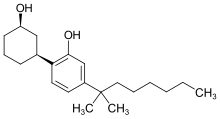
Cyclohexylphenols: Any compound containing a 2-(3-hydroxycyclohexyl)phenol structure with substitution at the 5-position of the phenolic ring by an alkyl, haloalkyl, alkenyl, cycloalkylmethyl, cycloalkylethyl, 1-(N-methyl-2-piperidinyl)methyl, or 2-(4-morpholinyl)ethyl group whether or not substituted in the cyclohexyl ring to any extent.
Some jurisdictions have addressed this by naming such compounds specifically, alternatively some have adopted broader definitions (such as in the Australian Federal Poisons Standard, which controls all derivatives of cyclohexylphenol unless otherwise specified).
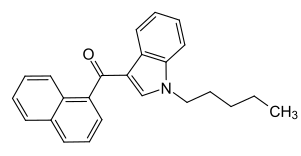
Notice the pentyl group on the nitrogen atom of the pyrrole ring of the molecule.
Strictly speaking this chemical name is incorrect, as JWH-176 and related compounds would more correctly be viewed as derivatives of 1-(1-naphthylmethylylidene)indene due to the unsaturated =CH- linker group (as opposed to the -CH2- linker group found in e.g. naphthylmethylindoles), however "Naphthylmethylindenes" has gained acceptance as a legal term of art in this instance.
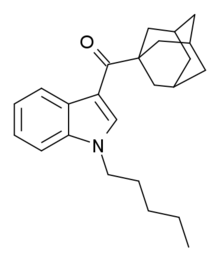
Indole-3-carboxylate esters: Any compound containing a 1H-indole-3-carboxylate ester structure with the ester oxygen bearing a napthyl, quinolinyl, isoquinolinyl, or adamantyl group and substitution at the one position of the indole ring by an alkyl, haloalkyl, alkenyl, cycloalkylmethyl, cycloalkylethyl, benzyl, N-methyl-2-piperidinylmethyl, or 2-(4-morpholinyl)ethyl group, whether or not further substituted on the indole ring to any extent and whether or not further substituted on the naphthyl, quinolinyl, isoquinolinyl, adamantyl, or benzyl groups to any extent.
One consequence of the introduction of broad Markush structure bans, has been the appearance of compounds which have similar structures but technically fall outside the scope of the legal definitions (for instance containing an indazole core instead of the proscribed indole, or a carboxamide linker in place of methanone), therefore resulting over time in a progressively increasing structural diversity of synthetic cannabinoids sold for illicit recreational use.
[24][25][26][27] Some jurisdictions have attempted to introduce a broader scope of coverage by defining "head", "core", "linker" and "tail" groups which can be interchanged in any combination that fits within the definition, resulting in a much wider (but still usually finite) range of compounds being encompassed.
[28] For example, the wording of this Texas statute encompasses a large range of "prophetic" core structures which have not yet been encountered in synthetic cannabinoids, but might plausibly be likely to appear in future (e.g. "Quinolinoyl pyrazole carboxylate", "Naphthoylimidazole" etc); "(a) In this section: (1) “Core component” is one of the following: azaindole, benzimidazole, benzothiazole, carbazole, imidazole, indane, indazole, indene, indole, pyrazole, pyrazolopyridine, pyridine, or pyrrole.
(3) “Link component” is one of the following functional groups: carboxamide, carboxylate, hydrazide, methanone (ketone), ethanone, methanediyl (methylene bridge), or methine... ...(5) any compound containing a core component substituted at the 1-position to any extent, and substituted at the 3-position with a link component attached to a group A component, whether or not the core component or group A component are further substituted to any extent"[29] Another approach (here from the UK) is to list an example structure and then specify ways in which it can be modified by swapping various parts of the molecule with alternative substituent groups, for example; "...any compound (not being clonitazene, etonitazene, acemetacin, atorvastatin, bazedoxifene, indometacin, losartan, olmesartan, proglumetacin, telmisartan, viminol, zafirlukast or a compound for the time being specified in sub-paragraphs (h) to (s) above) structurally related to 1-pentyl-3-(1-naphthoyl)indole (JWH-018), in that the four sub-structures, that is to say the indole ring, the pentyl substituent, the methanone linking group and the naphthyl ring, are linked together in a similar manner, whether or not any of the sub-structures have been modified, and whether or not substituted in any of the linked sub-structures with one or more univalent substituents and, where any of the sub-structures have been modified, the modifications of the sub-structures are limited to any of the following, that is to say - Broadly worded controls such as above may inadvertently include large numbers of compounds which merely happen to have some structural similarity, but do not have similar pharmacological effects to the prohibited cannabinoid drugs.