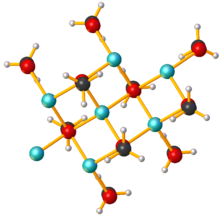
Sodium methoxide
The resulting solution, which is colorless, is often used as a source of sodium methoxide, but the pure material can be isolated by evaporation followed by heating to remove residual methanol.
[4] Sodium methoxide is a routinely used base in organic chemistry, applicable to the synthesis of numerous compounds ranging from pharmaceuticals to agrichemicals.
[6] Sodium methoxide is used as an initiator of anionic addition polymerization with ethylene oxide, forming a polyether with high molecular weight.
Sodium methoxide acts as a catalyst for this reaction, but will combine with any free fatty acids present in the oil/fat feedstock to form soap byproducts.
[citation needed] Commercial batches of sodium methoxide show variable levels of degradation, and were a major source of irreproducibility when used in Suzuki reactions.