Peptide deformylase
In enzymology, a peptide deformylase (EC 3.5.1.88) is an enzyme that removes the formyl group from the N terminus of nascent polypeptide chains in eubacteria, mitochondria and chloroplasts.
[1] Peptide deformylases are metaloenzymes monomers and bind a metal cofactor, typically Fe(II) or Zn, in an active site.
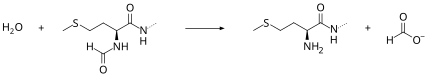
Peptide deformylase removes the formyl group from the N terminus of nascent polypeptides as they are synthesized by the ribosome.
The systematic name of this enzyme class is formyl-L-methionyl peptide amidohydrolase.
As of late 2007, 34 structures have been solved for this class of enzymes, with PDB accession codes 1IX1, 1LM4, 1LM6, 1LME, 1LQW, 1LQY, 1LRU, 1LRY, 1N5N, 1Q1Y, 1S17, 1SV2, 1SZZ, 1V3Y, 1VEV, 1VEY, 1VEZ, 1WS0, 1WS1, 1XEM, 1XEN, 1XEO, 1Y6H, 1ZXZ, 1ZY0, 1ZY1, 2AI7, 2AI8, 2AI9, 2AIA, 2AIE, 2EW5, 2EW6, and 2EW7.