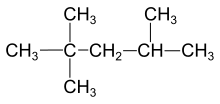
Single bond
When shared, each of the two electrons involved is no longer in the sole possession of the orbital in which it originated.
Rather, both of the two electrons spend time in either of the orbitals which overlap in the bonding process.
As a Lewis structure, a single bond is denoted as AːA or A-A, for which A represents an element.
The type of covalent bonding in hydrocarbons is extremely important in the nomenclature of these molecules.
Hydrocarbons containing only single bonds are referred to as alkanes (Moore, Stanitski, and Jurs 334).